THE NEW YORK TIMES
Just Spit and Wait- New Coronavirus Test Offers Advantage
Saliva Transport Stability:
 Saliva has not only been demonstrated to be a robust source of viral RNA but when preserved with Spectrum’s patented preservation solution it has been proven to protect COVID-19 transcripts for an extended period of time, making sample collection and transport to CLIA testing labs more efficient and amenable to global testing.
Saliva has not only been demonstrated to be a robust source of viral RNA but when preserved with Spectrum’s patented preservation solution it has been proven to protect COVID-19 transcripts for an extended period of time, making sample collection and transport to CLIA testing labs more efficient and amenable to global testing.
- The FDA specifies the SDNA-1000 as the ONLY device that is currently authorized for saliva collection for COVID-19 testing.
- “Saliva specimens must be collected, transported, and stored using the Spectrum Solutions LLC SDNA-1000 Saliva Collection Device.
- Rutgers’ Study proved that saliva samples can be shipped and stored at ambient temperatures with no degradation in quality.
- To support home use of the Spectrum Solutions LLC SDNA-1000 Saliva Collection Device, a Simulated Shipping Study was performed that was designed to evaluate the effect of temperature variation on the stability of SARS-CoV-2 RNA during transport of saliva specimens.
- The study was conducted using residual clinical specimens that had previously been reported as SARS-CoV-2 positive or negative and stored at -80°C until the start of the study. The SARS-CoV-2 positive specimens were selected based on the Ct values obtained upon initial testing and covered the spectrum of Ct values observed with the assay.
- The specimens were thawed and then subjected to various thermal profiles which were intended to simulate the extreme temperature conditions that may be experienced in the shipment of specimens during the summer and winter.
- At the conclusion of each thermal profile, the samples were retested with the Rutgers TaqPath SARS-CoV-2 Assay and the results were compared to those reported upon initial testing at the time the specimens were received.
- Nineteen out of 20 Low Positive samples (95%) and 10/10 High Positive samples were reported as positive after exposure to the summer and winter temperature excursions.
- The mean and standard deviation of the Ct values for each gene target was similar before and after simulated shipping, with no evidence of significant degradation of the SARS-CoV-2 RNA.
- All SARS-CoV-2 negative specimens were reported as “negative.”
- These results demonstrate that SARS-CoV-2 RNA positive saliva specimens are stable in the SDNA-1000 Saliva Collection Device when exposed to a broad range of temperature conditions.
- These data support the use of the SDNA-1000 Saliva Collection Device for transport and storage of specimens following home collection of saliva.
- The SDNA-1000 easily displayed its might in shipping simulations at extreme temperatures for over 10 days with no degradation of sample efficacy, proving sample stability and integrity.
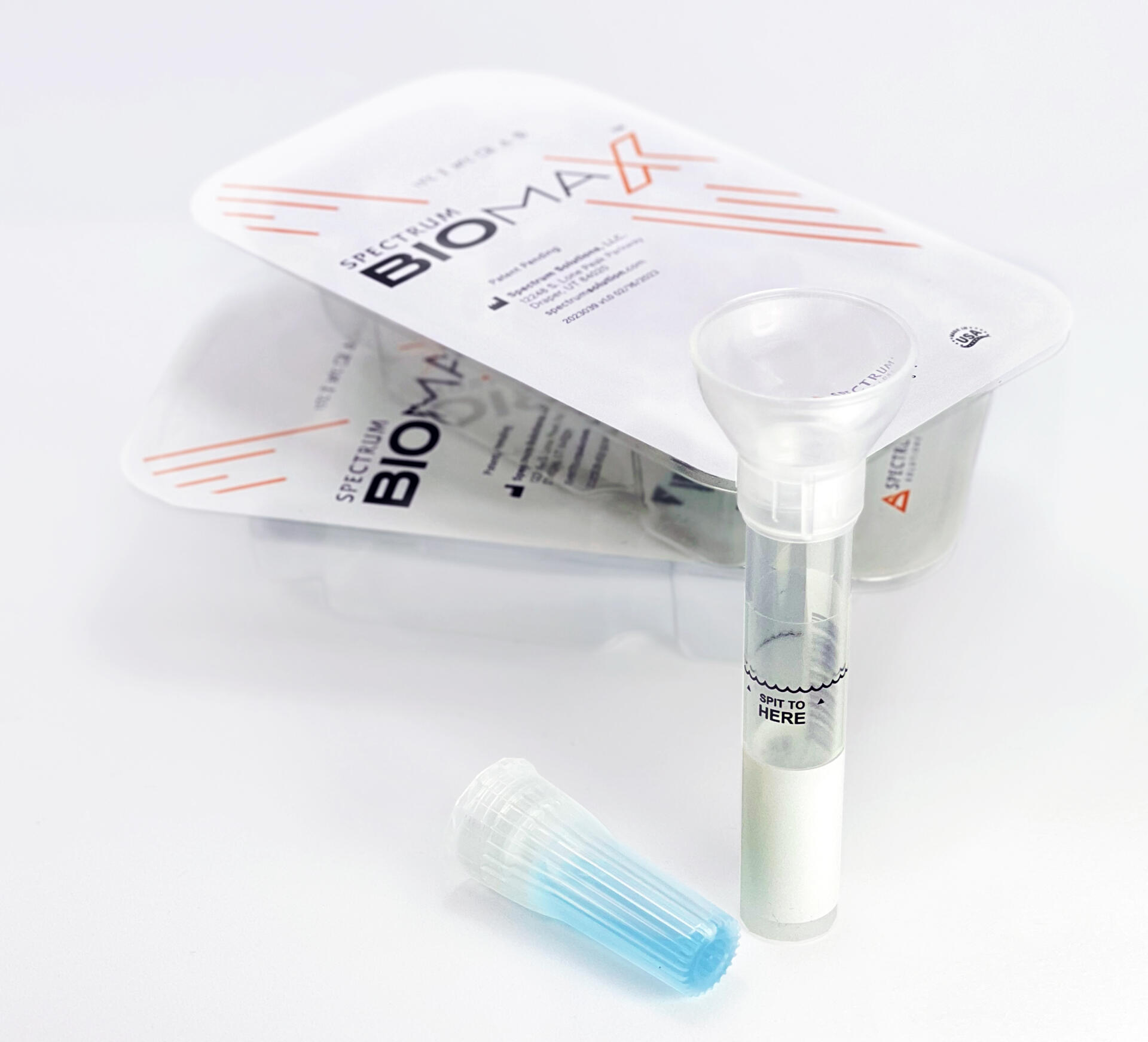
Noninvasive
Saliva Diagnostics
This changes everything!
Saliva analysis looks at the cellular level, the biologically active compounds, making it a true representative of what is clinically relevant. Engineered to lead the saliva collection industry, the BioMAX™ delivers the safest and most robust biosample for the earliest detection and diagnosis of disease and infection.
Since 2020 and the COVID-19 pandemic, Spectrum’s saliva collection system not only introduced, it continues to expand the molecular diagnostics industry and its understanding of the opportunities saliva offers patients, providers, and laboratories.
Noninvasive
Saliva Diagnostics
This changes everything!
Saliva analysis looks at the cellular level, the biologically active compounds, making it a true representative of what is clinically relevant. Engineered to lead the saliva collection industry, the BioMAX™ delivers the safest and most robust biosample for the earliest detection and diagnosis of disease and infection.
Since 2020 and the COVID-19 pandemic, Spectrum’s saliva collection system not only introduced, it continues to expand the molecular diagnostics industry and its understanding of the opportunities saliva offers patients, providers, and laboratories.
Bringing Baseball Back!
How you collect saliva makes a big difference
Increase workplace safety and build team confidence with simple and safe repeat testing programs supporting 100% accurate early detection and easy direct-to-user at-home options. Just ask Major League Baseball. See how our saliva collection system is credited for “bringing baseball back” and making Salt Lake “the league’s most important city”.
™/© 2020 MLB

Bringing Baseball Back!
How you collect saliva makes a big difference
Increase workplace safety and build team confidence with simple and safe repeat testing programs supporting 100% accurate early detection and easy direct-to-user at-home options. Just ask the Major League Baseball. See how our saliva collection system is credited for “bringing baseball back” and making Salt Lake “the league’s most important city”.
™/© 2020 MLB
About Spectrum Solutions®
Headquartered in Salt Lake City, Utah, Spectrum Solutions is dedicated to empowering complete wellness and bridging the gap between science and innovative healthcare solutions. Our stand-alone and fully integrated test-to-treat solutions support molecular diagnostics and DTC testing applications, advancing product development and accelerating go-to-market applications. Our single-source, end-to-end capabilities include a CAP/CLIA accredited molecular diagnostic laboratory, onsite compounding pharmacy, medical and non-medical product development, manufacturing, and fulfillment.
![]()
 Spectrum Corporate Spokesman
Spectrum Corporate Spokesman
Leslie Titus Bryant
Head of Marketing & Brand
admin@spectrumsolution.com
 Media Contact
Media Contact
Tim Rush, Springboard5
801-208-1100
tim.rush@springboard5.com
About Spectrum Solutions®
Headquartered in Salt Lake City, Utah, Spectrum Solutions is dedicated to empowering complete wellness and bridging the gap between science and innovative healthcare solutions. Our stand-alone and fully integrated test-to-treat solutions support molecular diagnostics and DTC testing applications, advancing product development and accelerating go-to-market applications. Our single-source, end-to-end capabilities include a CAP/CLIA accredited molecular diagnostic laboratory, onsite compounding pharmacy, medical and non-medical product development, manufacturing, and fulfillment.
![]()
 Spectrum Corporate Spokesman
Spectrum Corporate Spokesman
Leslie Titus Bryant
Head of Marketing & Brand
admin@spectrumsolution.com
 Media Contact
Media Contact
Tim Rush, Springboard5
801-208-1100
tim.rush@springboard5.com
Outside-of-the-Box Thinking, Inside-of-the-Box Innovation
Anywhere from customized testing solutions to new medical science product innovations–we’re here to help.




