FIRST EUA AUTHORIZED IVD DEVICE FOR SALIVA-BASED COVID-19 TESTING
Spectrum Solutions Secures Emergency Use Authorization (EUA) for SDNA-1000
Leslie Titus Bryant
October 08, 2020
New EUA Expanding Access and COVID-19 Testing Options
On October 8th, Spectrum Solutions received notice today that its SDNA-1000 saliva collection system had secured the first device U.S. Food and Drug Administration (FDA) EUA authorization for saliva-based COVID-19 testing. Spectrum’s self-contained saliva collection kit provides critical sample consistency while suspending and neutralizing viral RNA transcripts completely inactivating the live virus. Additionally, the SDNA-1000 stabilizes and protects saliva samples making both sample collection and transport for COVID-19 testing easy.
Spectrum and Rutgers Clinical Genomics Laboratory (RUCDR, now IBX) first identified, then introduced the nation to the key benefits of using saliva collected exclusively with the SDNA-1000 saliva collection system as a better viral diagnostic biomaterial for COVID-19 detection. Receiving its first EUA authorization from the FDA in March confirmed saliva’s tremendous diagnostic value and Spectrum’s innovative saliva collection device. COVID-19 saliva-based tests have already been sold and conducted using the SDNA-1000 under this previous EUA authorization held by RUCDR. However, this initial authorization limited broader access to saliva-based diagnostic testing, and restricted availability with only one testing platform ran by a single authorized lab.
First Saliva Collection System Authorized for At-Home Sample Self-Collection
This EUA authorization decision by the FDA successfully expands saliva-based testing access and availability through the validation of the SDNA-1000 saliva collection system on the broader range of platforms and assays currently being used by labs across the nation for immediate and seamless integration. Qualified commercial RNA extraction chemistries include Perkin Elmer, Thermo Fisher, Roche, and Qiagen.
“Saliva is a more robust biomaterial to facilitate molecular testing,” said Stephen Fanning, CEO of Spectrum Solutions. “Collected samples consistently demonstrate higher levels of testing accuracy, and the SDNA-1000 also offers the highest levels of safety by inactivating the live virus mitigating risks of exposure from the collection through shipping, to sample processing at the lab.”
Spectrum’s biomaterial collection system additionally provides:
- Mass-testing scalability and integration through validated and supported processes
- EUA authorized for over 10 days of post-collection stability for COVID-19 saliva samples with no degradation in sample efficacy
- EUA validated viral inactivation data
- First device authorized for at-home and unassisted sample self-collection
“The SDNA-1000 was engineered to innovate molecular diagnostic applications using saliva,” said Jeremy Johnson, VP of Engineering at Spectrum Solutions. “It’s exciting to offer labs the opportunity and freedom to explore additional DNA or viral RNA avenues for combating this pandemic and other viral infections with a safer biomaterial and a single biosample collection device.”
About Spectrum Solutions®
Headquartered in Salt Lake City, Utah, Spectrum Solutions is dedicated to empowering complete wellness and bridging the gap between science and innovative healthcare solutions. Our stand-alone and fully integrated test-to-treat solutions support molecular diagnostics and DTC testing applications, advancing product development and accelerating go-to-market applications. Our single-source, end-to-end capabilities include a CAP/CLIA accredited molecular diagnostic laboratory, onsite compounding pharmacy, medical and non-medical product development, manufacturing, and fulfillment.
![]()
 Spectrum Corporate Spokesman
Spectrum Corporate Spokesman
Leslie Titus Bryant
Head of Marketing & Brand
admin@spectrumsolution.com
 Media Contact
Media Contact
Tim Rush, Springboard5
801-208-1100
tim.rush@springboard5.com
About Spectrum Solutions®
Headquartered in Salt Lake City, Utah, Spectrum Solutions is dedicated to empowering complete wellness and bridging the gap between science and innovative healthcare solutions. Our stand-alone and fully integrated test-to-treat solutions support molecular diagnostics and DTC testing applications, advancing product development and accelerating go-to-market applications. Our single-source, end-to-end capabilities include a CAP/CLIA accredited molecular diagnostic laboratory, onsite compounding pharmacy, medical and non-medical product development, manufacturing, and fulfillment.
![]()
 Spectrum Corporate Spokesman
Spectrum Corporate Spokesman
Leslie Titus Bryant
Head of Marketing & Brand
admin@spectrumsolution.com
 Media Contact
Media Contact
Tim Rush, Springboard5
801-208-1100
tim.rush@springboard5.com
Bringing Baseball Back!
How you collect saliva makes a big difference
Increase workplace safety and build team confidence with simple and safe repeat testing programs supporting 100% accurate early detection and easy direct-to-user at-home options. Just ask Major League Baseball. See how our saliva collection system is credited for “bringing baseball back” and making Salt Lake “the league’s most important city”.
™/© 2020 MLB

Bringing Baseball Back!
How you collect saliva makes a big difference
Increase workplace safety and build team confidence with simple and safe repeat testing programs supporting 100% accurate early detection and easy direct-to-user at-home options. Just ask the Major League Baseball. See how our saliva collection system is credited for “bringing baseball back” and making Salt Lake “the league’s most important city”.
™/© 2020 MLB
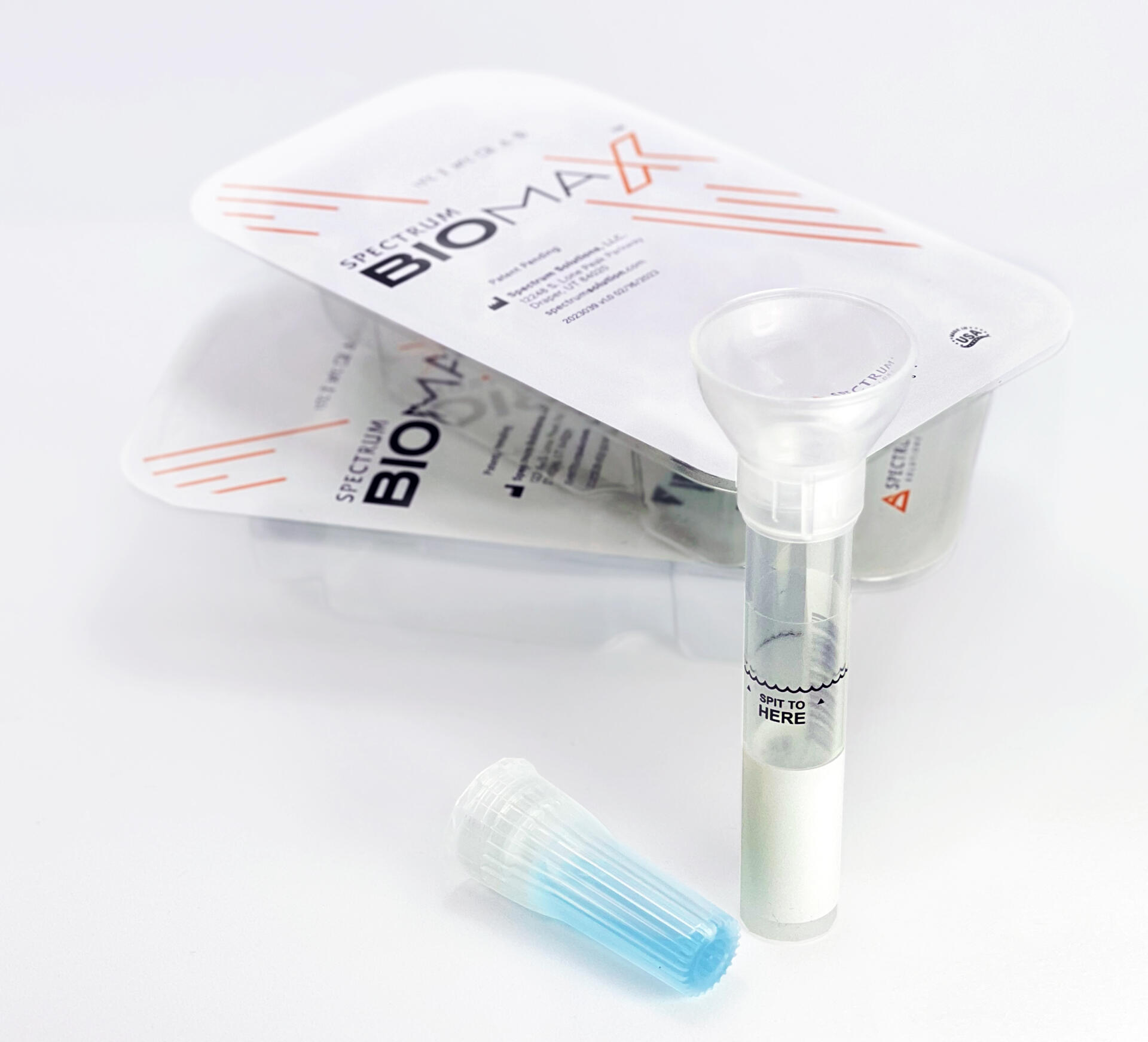
Noninvasive
Saliva Diagnostics
This changes everything!
Saliva analysis looks at the cellular level, the biologically active compounds, making it a true representative of what is clinically relevant. Engineered to lead the saliva collection industry, the BioMAX™ delivers the safest and most robust biosample for the earliest detection and diagnosis of disease and infection.
Since 2020 and the COVID-19 pandemic, Spectrum’s saliva collection system not only introduced, it continues to expand the molecular diagnostics industry and its understanding of the opportunities saliva offers patients, providers, and laboratories.
Noninvasive
Saliva Diagnostics
This changes everything!
Saliva analysis looks at the cellular level, the biologically active compounds, making it a true representative of what is clinically relevant. Engineered to lead the saliva collection industry, the BioMAX™ delivers the safest and most robust biosample for the earliest detection and diagnosis of disease and infection.
Since 2020 and the COVID-19 pandemic, Spectrum’s saliva collection system not only introduced, it continues to expand the molecular diagnostics industry and its understanding of the opportunities saliva offers patients, providers, and laboratories.
Outside-of-the-Box Thinking, Inside-of-the-Box Innovation
Anywhere from customized testing solutions to new medical science product innovations–we’re here to help.




