COVID-19: Rapid Antibody Screening & Saliva Testing
THE NEW YORK TIMES: Just Spit and Wait – New Coronavirus Test Offers Advantage
Press Release
Published: May 15, 2020
Published: May 15, 2020
The cities of Bluffdale, Draper, Riverton, and Vineyard are sponsoring COVID-19 antibody testing for city residents through a privately administered COVID-19 screening program. The goal of the program is to detect antibodies related to COVID-19. If individuals think they have been exposed, or just want to know if they have had COVID-19, they are invited to come and take a test.
Mayors from the participating communities released this joint statement about the project:
“We believe a central component to beating COVID-19 is to test our residents. We need to know our true infection rate so we can take actions to limit the spread, conduct contact tracing, and make future data-driven public policy decisions. We invite all our residents to consider being tested for COVID-19 antibodies as we continue to battle this virus.”
Any residents interested in being tested may sign up for an appointment online at rapidscreen.as.me, on a first-come, first-served basis. The screening costs $69.99 (may be reimbursable by health insurance plans), payable by credit or debit card at the time of booking. Appointments are booked in 30-minute increments throughout each day and conducted in a drive-thru setting. Results are provided within 15-20 minutes. The testing schedule is as follows:
- Wednesday, May 20 – Draper
- Thursday, May 21 – Riverton
- Tuesday, May 26 – Draper
- Thursday, May 28 – Bluffdale
- Friday, May 29 – Vineyard
Testing will be conducted by Utah-based RapidScreen Solutions, LLC (RSS) using testing kits manufactured in the United States from New York-based Chembio. The test is a lateral flow blood test that has Emergency Use Authorization (EUA) from the FDA. Using a finger-stick test, a small (drop) blood sample is taken from a finger and placed on a test strip to detect two different antibodies in the subject’s blood: IgM and IgG. Positive results indicate the likelihood of a current or past COVID-19 infection.

The cities of Bluffdale, Draper, Riverton, and Vineyard are sponsoring COVID-19 antibody testing for city residents through a privately administered COVID-19 screening program. The goal of the program is to detect antibodies related to COVID-19. If individuals think they have been exposed, or just want to know if they have had COVID-19, they are invited to come and take a test.
Mayors from the participating communities released this joint statement about the project:
“We believe a central component to beating COVID-19 is to test our residents. We need to know our true infection rate so we can take actions to limit the spread, conduct contact tracing, and make future data-driven public policy decisions. We invite all our residents to consider being tested for COVID-19 antibodies as we continue to battle this virus.”
Any residents interested in being tested may sign up for an appointment online at rapidscreen.as.me, on a first-come, first-served basis. The screening costs $69.99 (may be reimbursable by health insurance plans), payable by credit or debit card at the time of booking. Appointments are booked in 30-minute increments throughout each day and conducted in a drive-thru setting. Results are provided within 15-20 minutes. The testing schedule is as follows:
- Wednesday, May 20 – Draper
- Thursday, May 21 – Riverton
- Tuesday, May 26 – Draper
- Thursday, May 28 – Bluffdale
- Friday, May 29 – Vineyard
Testing will be conducted by Utah-based RapidScreen Solutions, LLC (RSS) using testing kits manufactured in the United States from New York-based Chembio. The test is a lateral flow blood test that has Emergency Use Authorization (EUA) from the FDA. Using a finger-stick test, a small (drop) blood sample is taken from a finger and placed on a test strip to detect two different antibodies in the subject’s blood: IgM and IgG. Positive results indicate the likelihood of a current or past COVID-19 infection.

FDA Authorized Saliva Testing will be used to Confirm Active COVID-19 Infection
In addition, RSS will randomly administer a saliva-based DNA test to some test subjects. Draper-based Spectrum Solutions manufactures the saliva tests that detect active COVID-19 infection, rather than detecting antibodies. Any person who tests positive for antibodies will also receive the saliva test to confirm active COVID-19 infection. The testing will be conducted in full compliance with FDA and CLIA requirements. The test results will not be diagnostic but are designed to provide valuable data about the COVID-19 infection rate. Check on the availability of diagnostic at-home COVID-19 testing options using saliva by visiting Spectrum’s website.

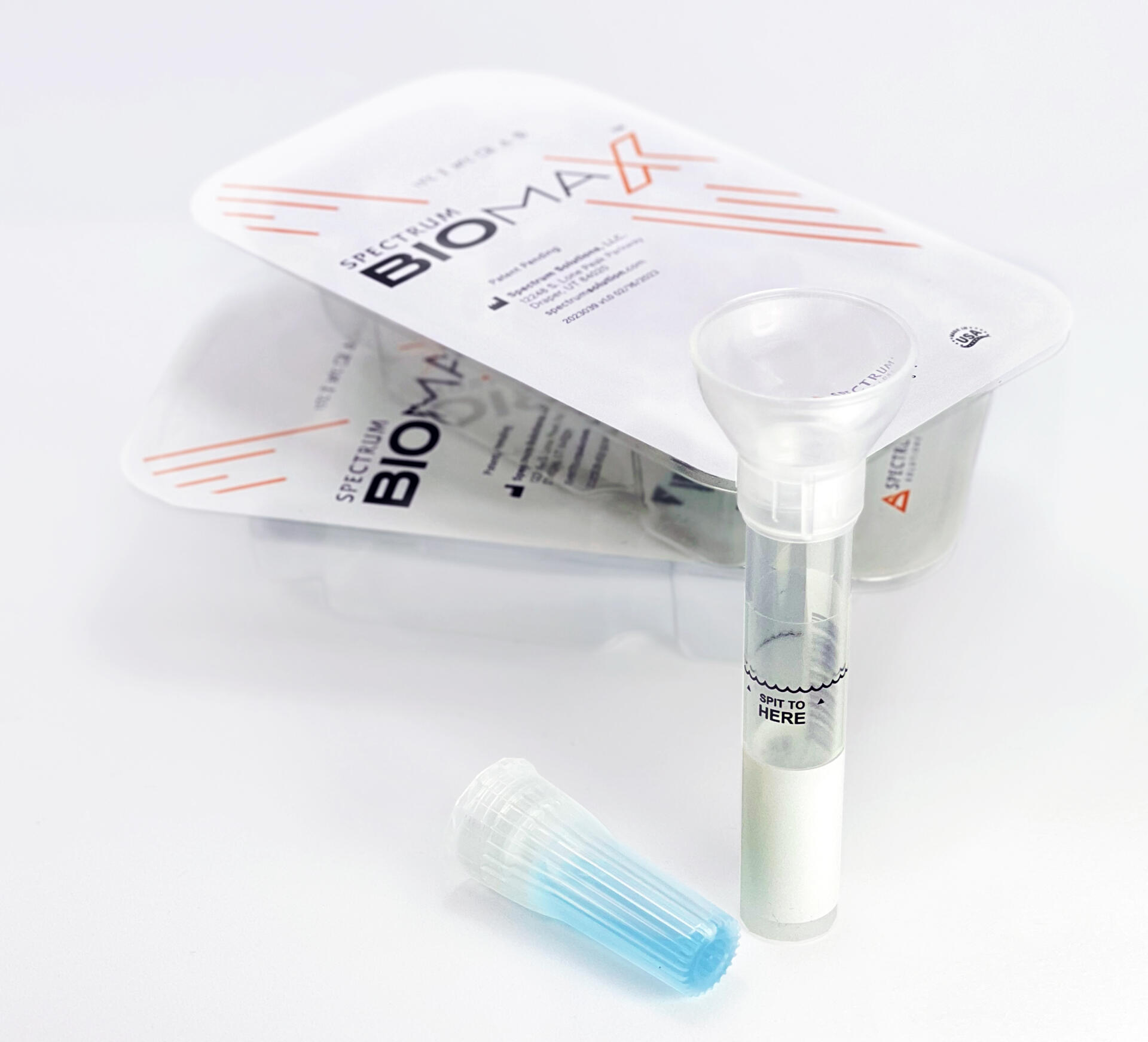
© Spectrum Solutions™ SDNA-1000 Only EUA Authorized Saliva Collection Device for COVID-19 Testing | Photo Credit: Leslie Titus Bryant
Spectrum
in the News
Spectrum
in the News
Noninvasive
Saliva Diagnostics
This changes everything!
Saliva analysis looks at the cellular level, the biologically active compounds, making it a true representative of what is clinically relevant. Engineered to lead the saliva collection industry, the BioMAX™ delivers the safest and most robust biosample for the earliest detection and diagnosis of disease and infection.
Since 2020 and the COVID-19 pandemic, Spectrum’s saliva collection system not only introduced, it continues to expand the molecular diagnostics industry and its understanding of the opportunities saliva offers patients, providers, and laboratories.
Noninvasive
Saliva Diagnostics
This changes everything!
Saliva analysis looks at the cellular level, the biologically active compounds, making it a true representative of what is clinically relevant. Engineered to lead the saliva collection industry, the BioMAX™ delivers the safest and most robust biosample for the earliest detection and diagnosis of disease and infection.
Since 2020 and the COVID-19 pandemic, Spectrum’s saliva collection system not only introduced, it continues to expand the molecular diagnostics industry and its understanding of the opportunities saliva offers patients, providers, and laboratories.
Outside-of-the-Box Thinking, Inside-of-the-Box Innovation
Anywhere from customized testing solutions to new medical science product innovations–we’re here to help.

