NPR (VIDEO):
What The U.S. Is Doing About Its Testing Swab Shortage
By: Sacha Pfeiffer and Meg Anderson
May 25, 2020
May 25, 2020

(3:18) Saliva Testing for COVID-19 – NPR
Widespread testing for the coronavirus is key to safely reopening the country, but the U.S. has struggled for months to get to the level of testing many experts say we need — even as states and cities begin to loosen restrictions.
Part of the problem is that a test for the coronavirus is not a single device. Testing entails several different steps and requires supplies and pieces of equipment sourced from different places. These supplies have to make it from factories to testing sites where patients’ samples can be collected and to the lab where tests are processed. Along the way, there can be shortages of various components, each one potentially creating a bottleneck. The most common kind of test to check for an active coronavirus infection takes a sample from the back of your nose or throat. There are dozens of varieties of that test, but they all require one key element: the swab. Not a painless solution as you can visually see and hear the “ooh, that doesn’t feel very good” come from the testing patients. Back to normal could mean repeated testing for many and painful swabbing a daily ritual. The only painless option is saliva and is currently FDA authorized using Spectrum’s SDNA Saliva Collection Device.
In this video, we zero in on the swab, detailing what the federal government and private industry did and didn’t do, the new saliva testing option using the Spectrum Solutions saliva collection device (spot 3:18 in the video), and we break down why testing is so complicated in the first place.
Spectrum
in the News
Spectrum
in the News
Noninvasive
Saliva Diagnostics
This changes everything!
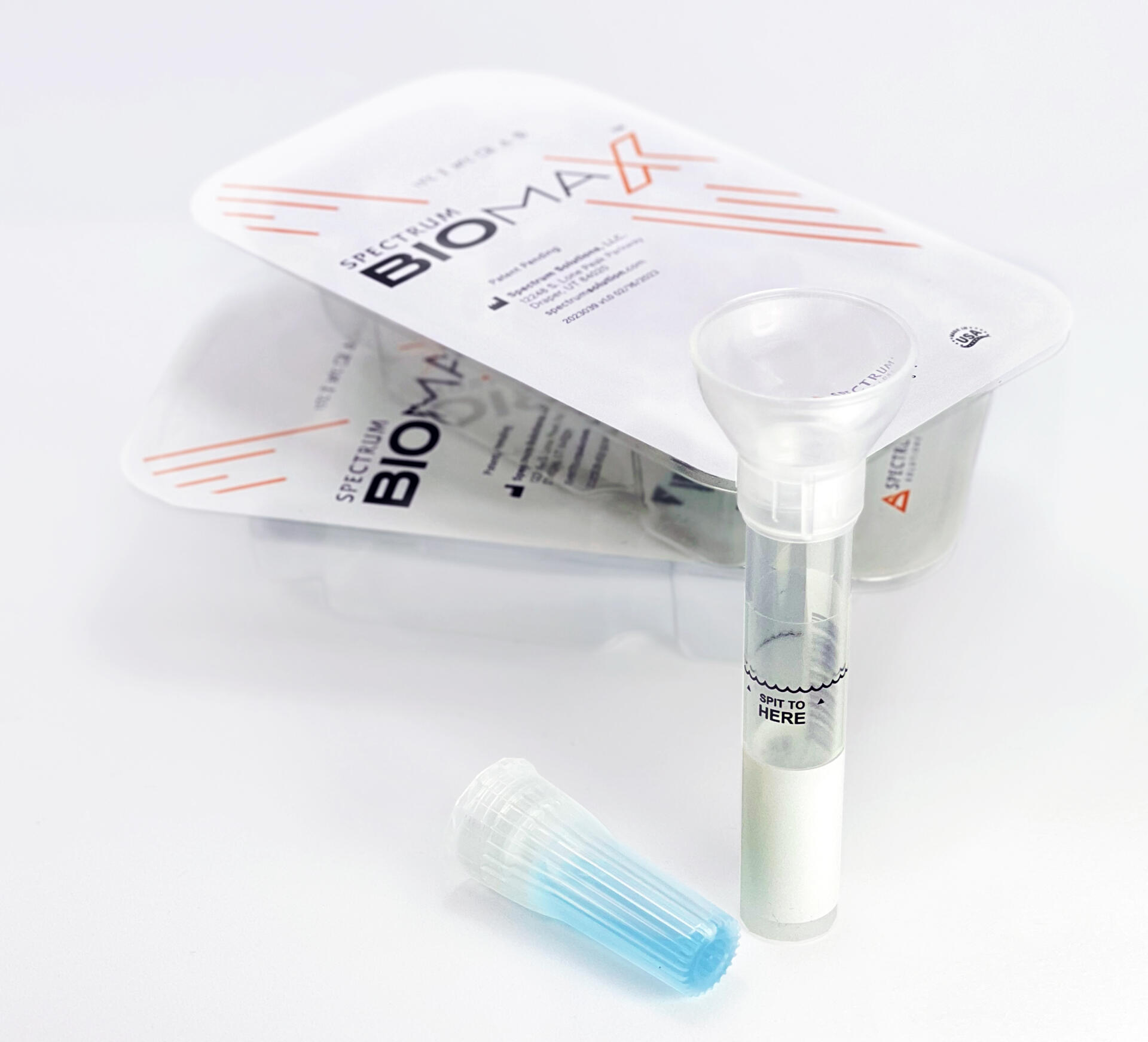
Saliva analysis looks at the cellular level, the biologically active compounds, making it a true representative of what is clinically relevant. Engineered to lead the saliva collection industry, the BioMAX™ delivers the safest and most robust biosample for the earliest detection and diagnosis of disease and infection.
Since 2020 and the COVID-19 pandemic, Spectrum’s saliva collection system not only introduced, it continues to expand the molecular diagnostics industry and its understanding of the opportunities saliva offers patients, providers, and laboratories.
Noninvasive
Saliva Diagnostics
This changes everything!
Saliva analysis looks at the cellular level, the biologically active compounds, making it a true representative of what is clinically relevant. Engineered to lead the saliva collection industry, the BioMAX™ delivers the safest and most robust biosample for the earliest detection and diagnosis of disease and infection.
Since 2020 and the COVID-19 pandemic, Spectrum’s saliva collection system not only introduced, it continues to expand the molecular diagnostics industry and its understanding of the opportunities saliva offers patients, providers, and laboratories.
Outside-of-the-Box Thinking, Inside-of-the-Box Innovation
Anywhere from customized testing solutions to new medical science product innovations–we’re here to help.