The COVID-19, 20, 21, 2022
Predictable Backslide
Leslie Titus Bryant
May 25, 2022
If we’ve learned anything from this COVID pandemic it should be:
- the value of early detection and the importance of accurate testing
- The fact that vaccinations actually don’t protect you from getting COVID if exposed to the virus (but do serve to lessen the disease severity)
- Those without a vaccination don’t spread COVID faster than anyone else
- Exposure doesn’t always become an active infection
- Every infection will share some, but not necessarily all the same symptoms
We’ve seen variants come and mutations go with consistent spikes in infection rates during the fall closely mirroring the pattern of behavior from another encapsulated RNA virus (the Flu) we have more experience with.
Each year, Flu season and colder weather make their way back around the calendar and with them come predictions from scientists estimating “over 100 million COVID cases and a potentially significant wave of deaths this fall and winter,” driven by new omicron subvariants that have shown a remarkable ability to escape immunity.
Getting the message out and understanding, as a population, the foundational importance of early detection and accurate testing will not only save lives but might be our only real chance at making this time around, the last time around.
The end of 2021 shined light on the new 2022 year with a “testing for everyone, vaccinated and unvaccinated” presidential mandate which at first appeared to be a huge step in the right direction. That is until details revealed that the testing they were speaking of was going to be rapid antigen testing, and then it wasn’t.
You’re not alone if you might be wondering why that is such a big deal. You see, most people don’t realize there are two types of technologies used to test for SARS-CoV-2. The first is RT-PCR testing and the second is Rapid Antigen Testing. The two types of tests use very different technology and look for very different things. RT-PCR testing (reverse transcription polymerase chain reaction) is considered a real-time diagnostic test using a virus’s genetic DNA material for accurate and proof positive detection. With over 99% accuracy this testing has always been and still is considered the gold standard with a 24-hour turnaround. Rapid antigen tests have been commonly used at point-of-care to detect active viral infections from influenza and RSV respiratory pathogens using protein fragments. Rapid tests get their name from being able to produce results quickly (within approximately 15–30 minutes). Like many respiratory viruses, it takes several days for COVID-19 to build up in your body after exposure. During this early phase of infection, the amount of viral protein is relatively low, and a rapid test may not detect your infection. Rapid antigen tests are most often accurate when a person is infectious because that is when the highest amount of virus is in the respiratory tract.
According to the CDC, “a person with COVID-19 is considered infectious starting 2 days before they develop symptoms, or 2 days before the date of their positive test if they do not have symptoms.” What does this mean and can we even detect an infection days before symptoms develop? Yes, not only can you detect an infection you can do it safely, (neutralizing the virus and mitigating spread) pain-free, and from work, home, or anywhere in between using saliva. It means the most important window of opportunity in preventing or mitigating exponentially spreading the virus is early detection, before symptoms present.
Saliva-based RT-PCR testing entered the scene early in April of 2020 from a commissioned study by Spectrum Solutions with Rutgers University. This study was the first to demonstrate the benefits of saliva-based diagnostics to the FDA for COVID-19 testing (receiving the first EUA saliva testing authorization) and was later supported by dozens of additional research studies from organizations and universities like Yale, Princeton, MIT, and others. Research studies that demonstrated the ability to accurately identify a positive COVID-19 infection using saliva 4-5 days before symptoms appear. This accurate detection was 2-3 days sooner than the previous RT-PCR gold standard test using a nasal swab.
The problem we have, according to Thermo Fisher Scientific’s “Get the Facts on PCR & Antigen Tests” data sheet, rapid antigen testing is at best only 64% accurate in symptomatic cases. Let that sink in for just a second. What does this mean? First, if you are symptomatic, you’ve already been spreading the disease for days. Second, this means 36% of positive cases are actually receiving a negative result and these individuals are going about their normal activities unknowingly putting everyone in their path at risk.
If the most important window of opportunity in preventing or mitigating exponentially spreading the virus is early detection, before symptoms present, then the rise in popularity of rapid antigen testing makes no sense at all. According to the data sheet, the case for early detection numbers speak loudly for themselves, “rapid antigen testing is only 36% accurate in asymptomatic (before symptoms present) cases”. If we do the math, this means 64% of actual positive cases receive a negative result from that convenient and super-fast antigen test.
The huge spikes we’ve seen each fall in positive COVID infections and those predicted to come for all of us (vaccinated and unvaccinated), can only be positively impacted by updating testing protocols to ensure the benefits of early detection from repeat RT-PCR screening as our first line of defense. The Thermo Fisher datasheet points out antigen testing does have a place. It’s just clearly not early detection and definitely not for asymptomatic populations. Simply look at the last two years and you can see when it’s outside of its intended place it has proven to be ineffective and will continue to place the greater population at risk. This earliest detection opportunity using samples collected with Spectrum’s saliva collection kit not only delivers the advance warning benefit needed to take effective quarantine action, it also safely neutralizes any live virus, in-device, at room temperature mitigating any risks of exposure for downstream processes. Successful early and accurate detection together with a 5-to-7-day isolation which includes being without a fever for 24 hours could be all we need to manage and effectively stop the spread.
The COVID-19, 20, 21, 2022
Predictable Backslide
Leslie Titus Bryant
May 25, 2022
If we’ve learned anything from this COVID pandemic it should be:
- the value of early detection and the importance of accurate testing
- The fact that vaccinations actually don’t protect you from getting COVID if exposed to the virus (but do serve to lessen the disease severity)
- Those without a vaccination don’t spread COVID faster than anyone else
- Exposure doesn’t always become an active infection
- Every infection will share some, but not necessarily all the same symptoms
We’ve seen variants come and mutations go with consistent spikes in infection rates during the fall closely mirroring the pattern of behavior from another encapsulated RNA virus (the Flu) we have more experience with.
Each year, Flu season and colder weather make their way back around the calendar and with them come predictions from scientists estimating “over 100 million COVID cases and a potentially significant wave of deaths this fall and winter,” driven by new omicron subvariants that have shown a remarkable ability to escape immunity.
Getting the message out and understanding, as a population, the foundational importance of early detection and accurate testing will not only save lives but might be our only real chance at making this time around, the last time around.
The end of 2021 shined light on the new 2022 year with a “testing for everyone, vaccinated and unvaccinated” presidential mandate which at first appeared to be a huge step in the right direction. That is until details revealed that the testing they were speaking of was going to be rapid antigen testing, and then it wasn’t.
You’re not alone if you might be wondering why that is such a big deal. You see, most people don’t realize there are two types of technologies used to test for SARS-CoV-2. The first is RT-PCR testing and the second is Rapid Antigen Testing. The two types of tests use very different technology and look for very different things. RT-PCR testing (reverse transcription polymerase chain reaction) is considered a real-time diagnostic test using a virus’s genetic DNA material for accurate and proof positive detection. With over 99% accuracy this testing has always been and still is considered the gold standard with a 24-hour turnaround. Rapid antigen tests have been commonly used at point-of-care to detect active viral infections from influenza and RSV respiratory pathogens using protein fragments. Rapid tests get their name from being able to produce results quickly (within approximately 15–30 minutes). Like many respiratory viruses, it takes several days for COVID-19 to build up in your body after exposure. During this early phase of infection, the amount of viral protein is relatively low, and a rapid test may not detect your infection. Rapid antigen tests are most often accurate when a person is infectious because that is when the highest amount of virus is in the respiratory tract.
According to the CDC, “a person with COVID-19 is considered infectious starting 2 days before they develop symptoms, or 2 days before the date of their positive test if they do not have symptoms.” What does this mean and can we even detect an infection days before symptoms develop? Yes, not only can you detect an infection you can do it safely, (neutralizing the virus and mitigating spread) pain-free, and from work, home, or anywhere in between using saliva. It means the most important window of opportunity in preventing or mitigating exponentially spreading the virus is early detection, before symptoms present.
Saliva-based RT-PCR testing entered the scene early in April of 2020 from a commissioned study by Spectrum Solutions with Rutgers University. This study was the first to demonstrate the benefits of saliva-based diagnostics to the FDA for COVID-19 testing (receiving the first EUA saliva testing authorization) and was later supported by dozens of additional research studies from organizations and universities like Yale, Princeton, MIT, and others. Research studies that demonstrated the ability to accurately identify a positive COVID-19 infection using saliva 4-5 days before symptoms appear. This accurate detection was 2-3 days sooner than the previous RT-PCR gold standard test using a nasal swab.
The problem we have, according to Thermo Fisher Scientific’s “Get the Facts on PCR & Antigen Tests” data sheet, rapid antigen testing is at best only 64% accurate in symptomatic cases. Let that sink in for just a second. What does this mean? First, if you are symptomatic, you’ve already been spreading the disease for days. Second, this means 36% of positive cases are actually receiving a negative result and these individuals are going about their normal activities unknowingly putting everyone in their path at risk.
If the most important window of opportunity in preventing or mitigating exponentially spreading the virus is early detection, before symptoms present, then the rise in popularity of rapid antigen testing makes no sense at all. According to the data sheet, the case for early detection numbers speak loudly for themselves, “rapid antigen testing is only 36% accurate in asymptomatic (before symptoms present) cases”. If we do the math, this means 64% of actual positive cases receive a negative result from that convenient and super-fast antigen test.
The huge spikes we’ve seen each fall in positive COVID infections and those predicted to come for all of us (vaccinated and unvaccinated), can only be positively impacted by updating testing protocols to ensure the benefits of early detection from repeat RT-PCR screening as our first line of defense. The Thermo Fisher datasheet points out antigen testing does have a place. It’s just clearly not early detection and definitely not for asymptomatic populations. Simply look at the last two years and you can see when it’s outside of its intended place it has proven to be ineffective and will continue to place the greater population at risk. This earliest detection opportunity using samples collected with Spectrum’s saliva collection kit not only delivers the advance warning benefit needed to take effective quarantine action, it also safely neutralizes any live virus, in-device, at room temperature mitigating any risks of exposure for downstream processes. Successful early and accurate detection together with a 5-to-7-day isolation which includes being without a fever for 24 hours could be all we need to manage and effectively stop the spread.
The Real Answer
Should be Pretty Clear
Any collective and proactive safety protocol should include repeat weekly saliva-based RT-PCR testing for its noninvasive, 99.998% accurate, and earliest detection advantage–over 4 days sooner than the nasal swab and before any symptoms are present. This time around, let’s get back to placing value on accuracy, understanding the importance of early asymptomatic detection, and saving lives. When the importance of all the facts sink in, it’s easy to understand saliva testing using the Spectrum Solution collection system with easy direct-to-patient at-home, at work, or even in-clinic testing options is not only critical, it’s crucial to actually obtaining accurate results for both asymptomatic and symptomatic cases.
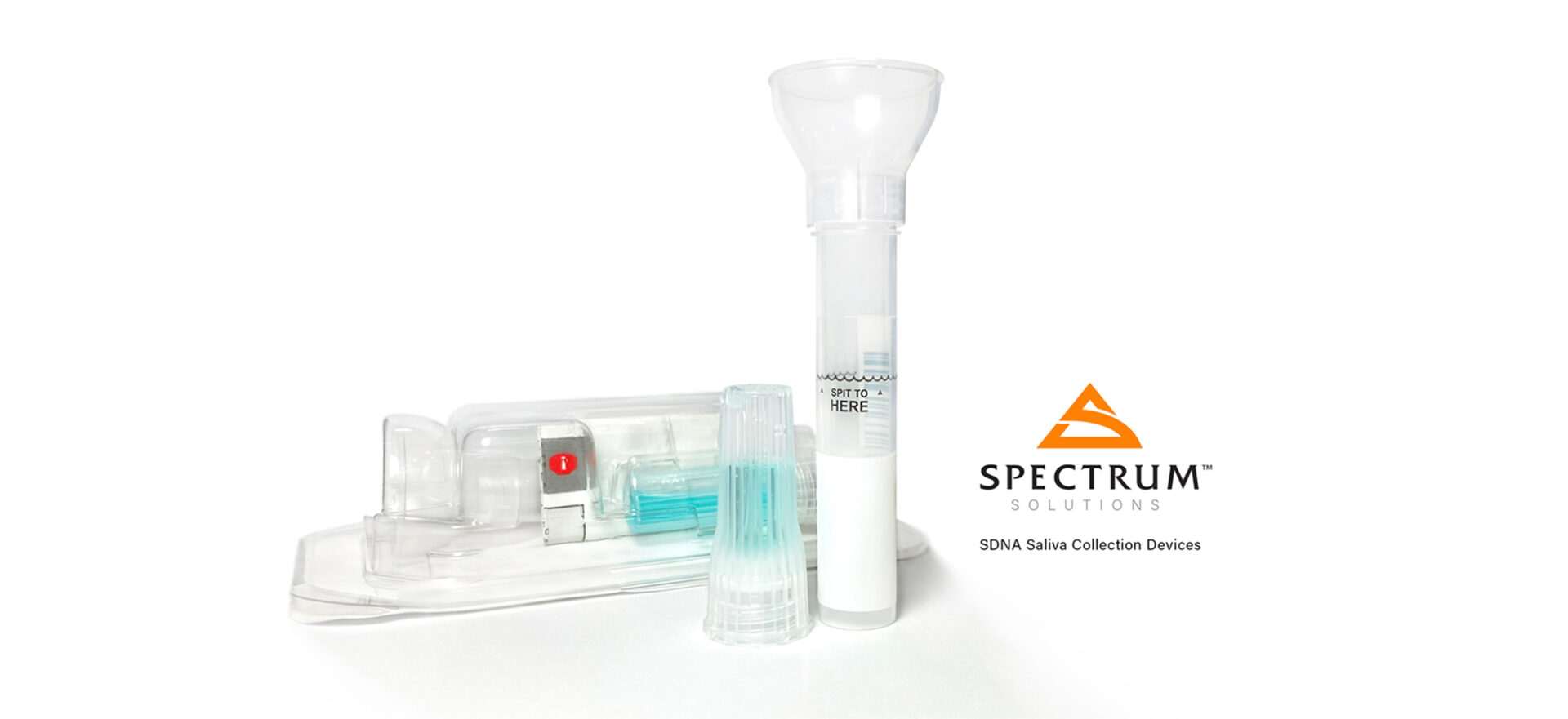 How you collect, preserve, and transport saliva is pivotal. As an in vitro diagnostic (IVD) medical device developer, Spectrum Solutions has engineered its innovative saliva collection system to eliminate sample self-collection errors delivering labs the most consistent, robust, and safest testing samples that provide the most accurate and earliest disease detection opportunity. Headquartered in Salt Lake City, Utah, Spectrum is a life science company using its focused industry expertise to drive noninvasive saliva molecular diagnostic and screening innovation. Our pioneering research, products, engineering, and IVD med-device manufacturing capabilities are setting new standards of modernized care for patients, providers, and laboratories in the testing, managing, and treatment of disease. When you bridge the gap between science and real medical solutions, you deliver innovation with the practical power to impact and change outcomes.
How you collect, preserve, and transport saliva is pivotal. As an in vitro diagnostic (IVD) medical device developer, Spectrum Solutions has engineered its innovative saliva collection system to eliminate sample self-collection errors delivering labs the most consistent, robust, and safest testing samples that provide the most accurate and earliest disease detection opportunity. Headquartered in Salt Lake City, Utah, Spectrum is a life science company using its focused industry expertise to drive noninvasive saliva molecular diagnostic and screening innovation. Our pioneering research, products, engineering, and IVD med-device manufacturing capabilities are setting new standards of modernized care for patients, providers, and laboratories in the testing, managing, and treatment of disease. When you bridge the gap between science and real medical solutions, you deliver innovation with the practical power to impact and change outcomes.
Ground-breaking benefits of saliva-based COVID-19 testing using Spectrum’s saliva collection kit:
- Noninvasive & pain-free specimen self-collection with the highest testing accuracy and earliest detection opportunity
- Identify infections at their earliest stage from as few as 200 copies/ml using patented preservation chemistry (as opposed to raw/neat saliva without preservation or lateral flow rapid tests needing 10,000-20,000 copies/ml)
- Increased access and test availability with first direct-to-patient at-home saliva testing FDA authorization
- At-home testing strengthens effective quarantine practices if positive, and eliminates any additional or unnecessary in-clinic exposure risks for families or individuals if negative
- Instant in-device live virus neutralization post-collection at room temperature
- Mitigates viral infection spread and exposure risks throughout the testing process for healthcare teams and downstream processing
- A greater than 90% reduction in the use/need for personal protective equipment (PPE)
- Engineered to eliminate user sample self-collection errors
- Provides the most consistent and robust testing sample with less variability for optimal test accuracy
- Able to pinpoint the lifecycle stage of active viral infection
- Delivers weeks of post-collection stability with no degradation of sample efficacy
- Removes the need for any temperature-controlled storage or transport of samples
- Delivers 99.998% testing accuracy & highest levels of test sensitivity
- Clinical testing commercial kitting services for mass-testing scalability & easy device integration
Major League Baseball Case Study
Practical application demonstrating Spectrum’s saliva testing
innovation and its credit for bringing baseball back.
“We would like to thank Spectrum Solutions for playing such an integral role in Major League Baseball’s return to the field in 2020. We needed a safe, reliable, and non-invasive way to regularly test and monitor players and Club personnel this season. Spectrum kit has proven to be the best option available for our ongoing needs. We appreciate Spectrum’s support, collaboration, and assistance during this challenging time.”
-Jon Coyles, VP Health & Safety for MLB
The Real Answer
Should be Pretty Clear
Any collective and proactive safety protocol should include repeat weekly saliva-based RT-PCR testing for its noninvasive, 99.998% accurate, and earliest detection advantage–over 4 days sooner than the nasal swab and before any symptoms are present. This time around, let’s get back to placing value on accuracy, understanding the importance of early asymptomatic detection, and saving lives. When the importance of all the facts sink in, it’s easy to understand saliva testing using the Spectrum Solution collection system with easy direct-to-patient at-home, at work, or even in-clinic testing options is not only critical, it’s crucial to actually obtaining accurate results for both asymptomatic and symptomatic cases.
 How you collect, preserve, and transport saliva is pivotal. As an in vitro diagnostic (IVD) medical device developer, Spectrum Solutions has engineered its innovative saliva collection system to eliminate sample self-collection errors delivering labs the most consistent, robust, and safest testing samples that provide the most accurate and earliest disease detection opportunity. Headquartered in Salt Lake City, Utah, Spectrum is a life science company using its focused industry expertise to drive noninvasive saliva molecular diagnostic and screening innovation. Our pioneering research, products, engineering, and IVD med-device manufacturing capabilities are setting new standards of modernized care for patients, providers, and laboratories in the testing, managing, and treatment of disease. When you bridge the gap between science and real medical solutions, you deliver innovation with the practical power to impact and change outcomes.
How you collect, preserve, and transport saliva is pivotal. As an in vitro diagnostic (IVD) medical device developer, Spectrum Solutions has engineered its innovative saliva collection system to eliminate sample self-collection errors delivering labs the most consistent, robust, and safest testing samples that provide the most accurate and earliest disease detection opportunity. Headquartered in Salt Lake City, Utah, Spectrum is a life science company using its focused industry expertise to drive noninvasive saliva molecular diagnostic and screening innovation. Our pioneering research, products, engineering, and IVD med-device manufacturing capabilities are setting new standards of modernized care for patients, providers, and laboratories in the testing, managing, and treatment of disease. When you bridge the gap between science and real medical solutions, you deliver innovation with the practical power to impact and change outcomes.
Ground-breaking benefits of saliva-based COVID-19 testing using Spectrum’s saliva collection kit:
- Noninvasive & pain-free specimen self-collection with the highest testing accuracy and earliest detection opportunity
- Identify infections at their earliest stage from as few as 200 copies/ml using patented preservation chemistry (as opposed to raw/neat saliva without preservation or lateral flow rapid tests needing 10,000-20,000 copies/ml)
- Increased access and test availability with first direct-to-patient at-home saliva testing FDA authorization
- At-home testing strengthens effective quarantine practices if positive, and eliminates any additional or unnecessary in-clinic exposure risks for families or individuals if negative
- Instant in-device live virus neutralization post-collection at room temperature
- Mitigates viral infection spread and exposure risks throughout the testing process for healthcare teams and downstream processing
- A greater than 90% reduction in the use/need for personal protective equipment (PPE)
- Engineered to eliminate user sample self-collection errors
- Provides the most consistent and robust testing sample with less variability for optimal test accuracy
- Able to pinpoint the lifecycle stage of active viral infection
- Delivers weeks of post-collection stability with no degradation of sample efficacy
- Removes the need for any temperature-controlled storage or transport of samples
- Delivers 99.998% testing accuracy & highest levels of test sensitivity
- Clinical testing commercial kitting services for mass-testing scalability & easy device integration
Major League Baseball Case Study
Practical application demonstrating Spectrum’s saliva testing
innovation and its credit for bringing baseball back.
“We would like to thank Spectrum Solutions for playing such an integral role in Major League Baseball’s return to the field in 2020. We needed a safe, reliable, and non-invasive way to regularly test and monitor players and Club personnel this season. Spectrum kit has proven to be the best option available for our ongoing needs. We appreciate Spectrum’s support, collaboration, and assistance during this challenging time.”
-Jon Coyles, VP Health & Safety for MLB
Bringing Baseball Back!
How you collect saliva makes a big difference
Increase workplace safety and build team confidence with simple and safe repeat testing programs supporting 100% accurate early detection and easy direct-to-user at-home options. Just ask Major League Baseball. See how our saliva collection system is credited for “bringing baseball back” and making Salt Lake “the league’s most important city”.
™/© 2020 MLB

Bringing Baseball Back!
How you collect saliva makes a big difference
Increase workplace safety and build team confidence with simple and safe repeat testing programs supporting 100% accurate early detection and easy direct-to-user at-home options. Just ask the Major League Baseball. See how our saliva collection system is credited for “bringing baseball back” and making Salt Lake “the league’s most important city”.
™/© 2020 MLB
About Spectrum Solutions®
Headquartered in Salt Lake City, Utah, Spectrum Solutions is dedicated to empowering complete wellness and bridging the gap between science and innovative healthcare solutions. Our stand-alone and fully integrated test-to-treat solutions support molecular diagnostics and DTC testing applications, advancing product development and accelerating go-to-market applications. Our single-source, end-to-end capabilities include a CAP/CLIA accredited molecular diagnostic laboratory, onsite compounding pharmacy, medical and non-medical product development, manufacturing, and fulfillment.
![]()
 Spectrum Corporate Spokesman
Spectrum Corporate Spokesman
Leslie Titus Bryant
Head of Marketing & Brand
admin@spectrumsolution.com
 Media Contact
Media Contact
Tim Rush, Springboard5
801-208-1100
tim.rush@springboard5.com
About Spectrum Solutions®
Headquartered in Salt Lake City, Utah, Spectrum Solutions is dedicated to empowering complete wellness and bridging the gap between science and innovative healthcare solutions. Our stand-alone and fully integrated test-to-treat solutions support molecular diagnostics and DTC testing applications, advancing product development and accelerating go-to-market applications. Our single-source, end-to-end capabilities include a CAP/CLIA accredited molecular diagnostic laboratory, onsite compounding pharmacy, medical and non-medical product development, manufacturing, and fulfillment.
![]()
 Spectrum Corporate Spokesman
Spectrum Corporate Spokesman
Leslie Titus Bryant
Head of Marketing & Brand
admin@spectrumsolution.com
 Media Contact
Media Contact
Tim Rush, Springboard5
801-208-1100
tim.rush@springboard5.com




